MammaPrint+BluePrint欣扶妳乳癌腫瘤基因雙效檢測
通過美國FDA(食品藥物管理局)MammaPrint獲得五項認證
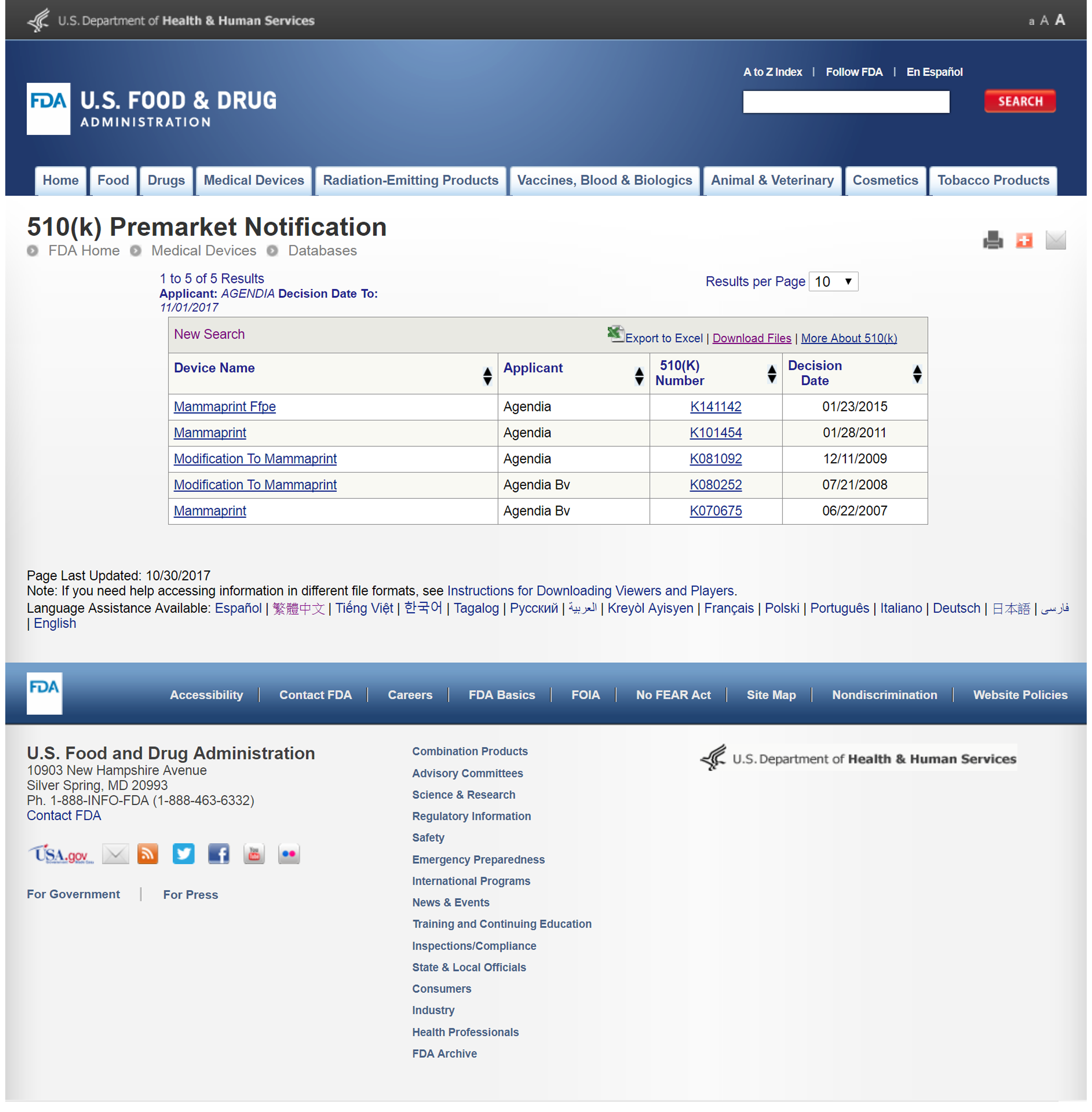
美國食品藥物管理局FDA在2007年批准MammaPrint欣扶妳乳癌腫瘤基因檢測,使用在乳癌患者的確診病例手術後的檢體做檢測,來預測乳癌是否會在5或10年內復發。

(認證編號:K101454、K080252、K070675、K081092、K141142)
1. 用乳癌腫瘤基因表現來評估乳癌復發與遠端轉移風險的體外診斷檢測
2. 美國加州與荷蘭阿姆斯特丹兩大實驗室品質管制,基因晶片合併DNA及RNA品質鑑定符合基因檢測標準
3. 高密度微陣列掃描技術
4. 新鮮檢體RNARetain保存技術
5. 突破過去的檢測年齡限制,檢測年齡最高可達87歲
6. FFPE(福爾馬林固定石蠟包埋檢體)可應用於MammaPrint分析70個基因乳癌復發風險檢測